News

The general module run 76, breast cancer module run B41 and HER2 ISH module run H29 showed generally high pass rates, with several markers demonstrating clear improvement compared with previous assessments. Despite logistical challenges related to slide shipment and delayed returns, sufficient material was returned in time to provide a reliable overview of assay performance. All the slides returned to NordiQC were assessed. Click on the underlined epitope name below to access the complete and detailed assessment report for each evaluated marker.
General module
Calretinin showed improved performance, with 85% of 408 laboratories achieving a sufficient result. The highest pass rate was obtained with Ready-To-Use systems from Ventana/Roche and Leica Biosystems when used according to vendor recommendations. The main challenge remained detection of low-level calretinin expression, particularly in granulosa cell tumour, adrenal cortical cells and adipocytes. Adipocytes in the submucosa of the appendix may serve as a useful and more accessible low-expressing control tissue.
CDX2 remained one of the strongest performing markers with 97% of 404 laboratories achieving a sufficient result. The high pass rate was supported by widespread use of robust clones such as EP25, EPR2764Y and DAK-CDX2, particularly in Ready-To-Use systems. Pancreas remains an important positive control for monitoring analytical sensitivity to low-level CDX2 expression.
CK5 was more challenging with only 69% of 330 laboratories achieving a sufficient result. The most frequent cause of insufficient results was the use of the less successful clone D5/16 B4. Ready-To-Use systems based on XM26 and SP27 performed best. Most laboratories were able to demonstrate CK5 in strongly positive tissues, whereas adequate staining of structures with lower CK5 expression, particularly mesothelioma and pancreatic intercalated ducts, was more challenging.
MLH1 showed clear improvement, with 90% of 373 laboratories achieving a sufficient result. The most common cause of insufficient results was weak or absent staining in cells expected to be positive. Detection of MLH1 in low level antigen expressing cells, especially stromal cells serving as internal controls in tumour samples, remains critical for reliable interpretation. The results also suggested that antibody lot age and stability may influence performance in some Ready-To-Use systems.
PAX5 also performed well overall with 91% of 335 laboratories achieving a sufficient result. Clone DAK-PAX5 showed the most robust performance, while clone SP34 was associated with aberrant staining in a substantial proportion of assays. The main technical challenge was achieving sufficient sensitivity to detect weak nuclear staining in Reed-Sternberg cells without introducing excessive aberrant staining in background.
Breast cancer module
ER recovered from the reduced performance seen in recent runs. In run B41, 91% of 400 laboratories achieved a sufficient result, with 61% assessed as optimal. The strongest performance was still observed with the Dako/Agilent Omnis system based on EP1. The Ventana/Roche SP1 Ready-To-Use system returned to expected performance after the previous problems related to recalled antibody lots. Low analytical sensitivity remained the main cause of insufficient results, underlining the continued importance of tonsil and uterine cervix as control tissues.
For HER2 IHC, 91% of 431 laboratories achieved a sufficient result. Companion diagnostic assays again performed better than laboratory developed tests. The HercepTest GE001 assay on Dako Omnis showed the strongest performance, with a 100% pass rate and 91% optimal results when used according to vendor recommendations. Ventana/Roche 4B5-based assays also performed well. While concordance for classical HER2 overexpression was high, HER2-low classification remained more demanding, with many “Good” results reflecting reduced sensitivity for HER2-low positive tumours.
HER2 ISH module
For HER2 ISH run H29, 158 laboratories participated in the technical BRISH assessment and 91% achieved a sufficient result, representing the highest pass rate observed to date. The improvement was partly related to revised assessment criteria introduced in run H24, allowing large negative areas when reliable HER2/chromosome 17 evaluation was still possible. The VENTANA Gene Protein Assay (GPA) showed improved performance compared with run H28. In the interpretative assessment, 230 laboratories submitted scoring sheets. Overall consensus was 79%, with higher concordance for FISH than for BRISH. This highlights that technical sufficiency does not necessarily ensure full reproducibility of the final diagnostic conclusion.
Overall, this assessment round demonstrated strong performance for several markers. At the same time, the results continued to identify important limitations related to analytical sensitivity, clone selection, signal-to-noise ratio and interpretative reproducibility. These findings underline the value of external quality assessment in detecting challenging IHC assays and monitoring quality.
The most recent NordiQC assessments covered the General module (Run 75), the Breast module (Run B40), the HER2 ISH module (Run H28) and the Companion module (Run C18). Below is a brief summary of the observed results. Click on the epitope name on the NordiQC website to access the complete assessment report for each marker, including best practice protocols and recommended immunohistochemical critical assay performance controls (iCAPCs).
As specified previously, we encourage participants to update submitted protocol settings and reagents if changed from the initially entered data. The submitted data are essential to analyse overall performance and identify root causes for both inferior and optimal performance.
General module - Run 75
AMACR: 379 laboratories participated and 92% achieved a sufficient result. Two rmAb clones dominated the assessment, 13H4 and SP116. The Dako/Agilent RTU assays based on clone 13H4 and Ventana/Roche RTU assays based on clone SP116 both achieved 100% sufficient results. However, for SP116-based assays, the proportion of optimal results was strongly influenced by choice of detection system, with markedly higher optimal rates observed when a 3-step detection system was applied.
CyD1: 407 laboratories participated and a pass rate of 95% was obtained, continuing the long-term improvement observed for this marker. The high pass rate was closely associated with the widespread use of robust clones (EP12 and SP4/SP4-R) and an extended shift towards standardised and robust RTU assays.
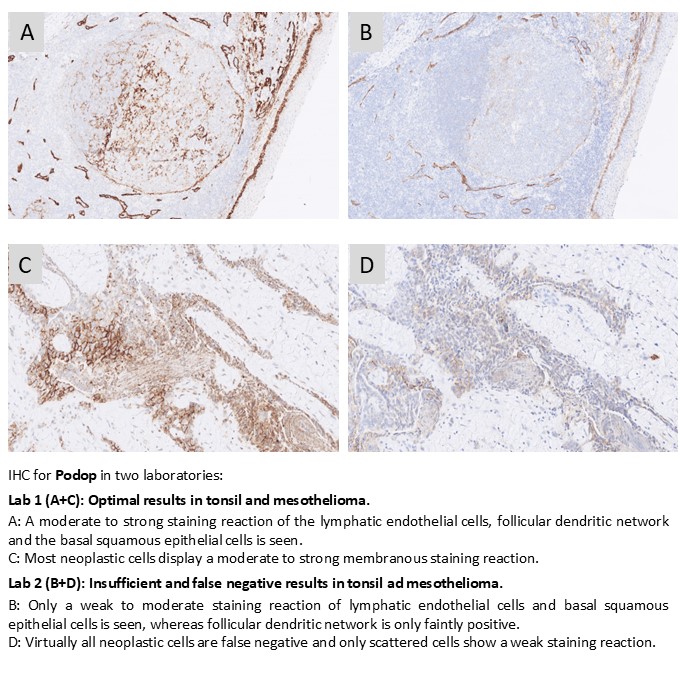
Podop: 373 laboratories participated and a pass rate of 72% was obtained. mAb clone D2-40 was used by 98% of participants. A major observation was the pronounced performance difference between Ready-To-Use (RTU) systems when applied strictly according to vendor recommended protocol settings. The Dako/Agilent IR072 RTU assay achieved a pass rate of 100% when used as recommended, whereas the Ventana/Roche RTU assay (760-4395) reached only 44% sufficient results under vendor recommended conditions. Importantly, optimisation of the Ventana protocol by changing from a 2-step UltraView detection system to a more sensitive 3-step OptiView system resulted in a substantial improvement, with the pass rate increasing to approximately 86%.
SATB2: 272 laboratories participated and a pass rate of 74% was obtained. Performance was highly dependent on antibody selection and associated protocol optimisation. Approximately 40% of the insufficient results were related to the use of less successful antibodies that failed to provide the expected staining results. The dominant rmAb clone EP281 provided the most robust results overall, but required careful adjustment of protocol settings, including the use of 3-step detection for concentrated formats, to achieve sufficient results. EP281 gave an inferior performance on Bond IHC platforms with no optimal results provided. In contrast the Ventana/Roche RTU system also based on EP281 was most successful with a pass rate of 89% when applied by vendor recommended conditions.
SOX10: 416 laboratories participated and a pass rate of 92% was obtained, representing a marked improvement compared with the first NordiQC assessments. The transition from polyclonal to monoclonal antibodies and the increasing use of standardised protocols were central to this development. The Ventana/Roche RTU assay based on clone SP267 showed particularly strong performance, achieving 100% sufficient and 96% optimal results.
Breast module - Run B40
Estrogen receptor (ER): 427 laboratories participated and a relatively low pass rate of 71% was obtained. Insufficient results were predominantly characterised by reduced analytical sensitivity, with weak or false negative nuclear staining reactions. RTU IHC assays were used by 94% of the participants. The low pass rate in this run was in particular caused by an inferior performance of the Ventana/Roche IHC assays based on rmAb clone SP1. These assays were used by 61% of all participants with an overall pass rate of only 67%. The vast majority of the participants used a lot of the RTU primary SP1 Ab being recalled by Roche in the period of this assessment run due to quality issues. The assessment further highlighted the importance of a continuous surveillance of IHC assays using reliable positive controls to monitor reproducibility of these.
HER2 IHC: 410 laboratories participated and a pass rate of 89% was obtained. Companion diagnostic (CDx) assays again showed superior performance. The HercepTest™ GE001 for Dako Omnis achieved a 100% pass rate with 76% optimal results when used according to vendor recommended protocol settings. Ventana/Roche HER2 IHC assays based on clone 4B5 reached an overall pass rate of 94%, and 61% of results being classified as optimal. An important finding was that many assays were successful for HER2 classical overexpression but showed a decreased agreement and concordance for HER2 Low compared to the level expected and defined by the NordiQC reference methods. This highlights the need to address an improved reproducibility for HER2 Low testing and a need to identify reliable control material to monitor precision and accuracy for HER2 IHC.
HER2 ISH module - Run H28
HER2 ISH: 174 laboratories participated in the technical assessment and an overall pass rate of 84% was obtained. The majority of participants used the VENTANA HER2 Dual ISH assay, which remained the most robust and consistent method. In contrast, the Gene Protein Assay (combined HER2 IHC and ISH) achieved a lower pass rate of 58%, likely related to impaired probe penetration in cases with dense IHC signal. In the scoring module, 219 laboratories submitted scoring sheets, and agreement with NordiQC reference diagnoses reached 87%. For laboratories performing FISH, the overall agreement rate was 91% compared to 86% for laboratories using BRISH.
Companion module - Run C18
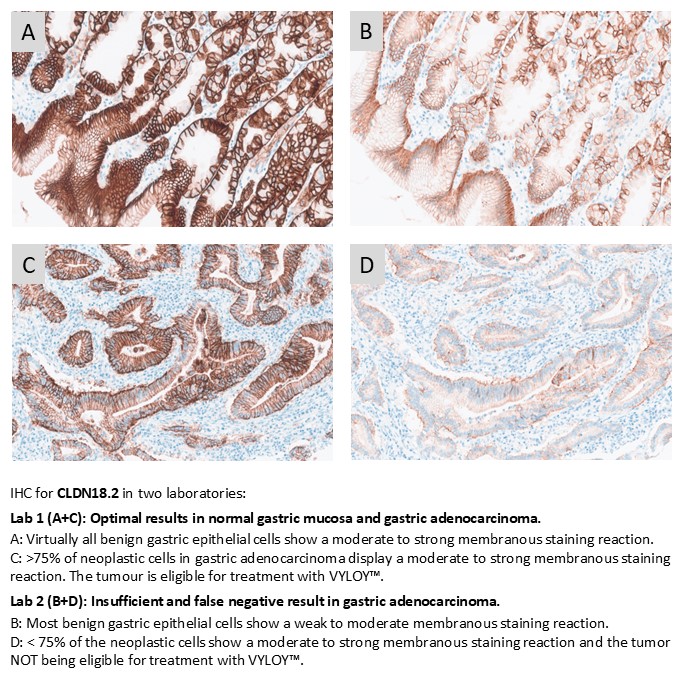
CLDN18.2: 90 laboratories participated and a pass rate of 92% was obtained, representing an improvement compared with the previous assessment. Ventana/Roche assays based on clone 43-14A, including the RxDx assay being used as reference standard method by NordiQC, showed the most robust performance, with 100% sufficient results and a high proportion of optimal results. Laboratory developed assays based on concentrated antibodies achieved a lower overall pass rate of 69%. Challenges related to selection of appropriate low-level tissue controls were noted.
PD-L1 TPS/CPS (KEYTRUDA®): 288 laboratories participated and a pass rate of 94% was obtained. Companion diagnostic assays continued to set the quality benchmark. The Dako/Agilent 22C3 GE006 assay achieved a 100% pass rate with 98% optimal results when used in accordance with vendor recommended protocol settings. The Ventana/Roche SP263 IHC assay also showed a high level of performance, reaching a pass rate of 97% when applied according to vendor recommendations, although the proportion of optimal results remained lower. Among laboratory developed tests, assays based on clone 22C3 in concentrated format achieved their highest pass rate to date at 92%, reflecting a marked improvement compared with previous runs. Insufficient results were mainly characterised by reduced proportions of PD-L1 positive cells compared with NordiQC reference levels, leading to false negative results.
December 18th 2025
Søren Nielsen
Scheme director
Run 74 (General module) and C17 (Companion module) were completed between April and July 2025. In total, approximately 600 laboratories from more than 50 countries participated in at least one of the two offered modules. Claudin 18.2 (CLDN18.2) was included as pilot run in in the Companion module run C17 for the first time.
Below is a summary of the observed results. Click on the epitope name to access the complete and detailed assessment report for each evaluated marker. Each report outlines best practice protocols with recommended immunohistochemical critical assay performance controls (iCAPCs) to support both the setup of IHC protocols and the ongoing surveillance of laboratory performance.
As specified previously, we do encourage participants to update the submitted protocol settings and reagents used if changed from initial entered data. Protocols can be updated until the assessment of the individual run. The submitted data are of central importance to analyze the overall performance and identify root causes for both inferior and optimal performance.
General module - run 74
BRAF: 211 laboratories participated and obtained a pass rate of 81% (56% optimal). The Ventana/Roche Ready-To-Use (RTU) system based on the mAb clone VE1 being used by 74% of all participants was the most successful assay with a pass rate of 97% when used accordingly to package insert. The corresponding concentrated format of clone VE1 applied within a laboratory developed (LD) assay provided an inferior performance and especially challenged on the Leica Biosystems Bond IHC platforms.
CD30: 423 laboratories participated and obtained a pass rate of 75% (39% optimal) similar to the level in the latest run 65, 2022. Both the widely used mAb clone Ber-H2 and clone JCM182 could be used to obtain an optimal staining result. The mAb clone Ber-H2 was found challenging on the Leica Biosystems Bond IHC platforms. 78% of the participants used RTU systems for CD30, being an increase from 70% in run 65. It was observed, that the RTU systems from the two main providers (Ventana/Roche and Dako/Agilent) provided an inferior performance when applied accordingly to package insert.
CLDN4: 97 laboratories participated in this first assessment for CLDN4 and obtained a pass rate of 72% (29% optimal). The assessment focused on differentiation of mesothelioma and adenocarcinoma using CLDN4. The rmAb clone EP417 was most successful for the demonstration of CLDN4. As concentrated format within a LD assay, optimal results were obtained on the stainer platforms from Ventana/Roche and Dako/Agilent. No RTU systems from the main IHC providers were available.
ECAD: 430 laboratories participated and obtained a pass rate of 76% (58% optimal) being reduced compared to the level of 89% in run 53, 2018. The inferior performance was highly impacted on the extensive use of less successful Abs as mAb clone 36B5 and the rmAb clones EP6 and EP700Y. In total 18% (n=78) of all submitted protocols were based on one of these three Abs giving a pass rate of only 8%. In contrast protocols based on mAb clone NCH-36 gave a pass rate of 95% (n=192) irrespectively of protocol settings applied.
MLA: 384 laboratories participated and a very low pass rate of only 34% was obtained and significantly declined to the nine previous assessments (mean 61%, range 32-88%). The assessment focused on the identification of malignant melanomas in the characterization of tumours of unknown origin. The mAb clone A103 was most frequently used by a total of 90% of the participants. However, only 26% of the protocols based on this clone provided a sufficient result. Insufficient results were typically characterized as too weak or false negative results. In contrast, the clones EP43 and BS52 gave a pass rate of 100%. The extensive difference in pass rates for the Abs compared sincerely underlines the need for both laboratories and vendors of IHC systems to optimize, validate and apply the most robust and effective IHC assays to secure diagnostic precision and reduce the risk of misdiagnosis.
Due to the low pass rate, MLA will be repeated in 2026 and no reassessment will be available.
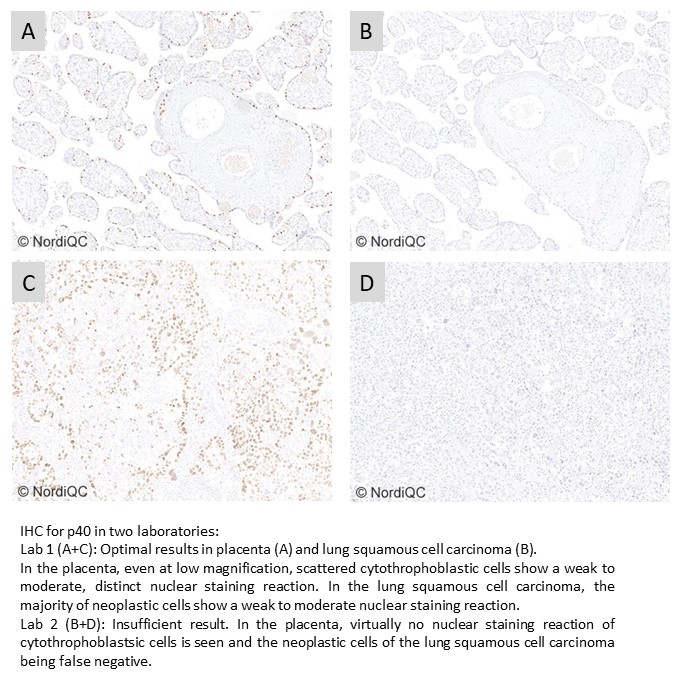
p40: 411 laboratories participated and obtained a pass rate of 94% (60% optimal) being the highest level for p40 to date. The assessment focused on p40 to differentiate lung adenocarcinoma and squamous cell carcinoma. The positive development was both related to the robust performance of the widely used mAb clone BC28 providing optimal results on all main IHC platforms but also to the access of successful and well-calibrated RTU systems from Ventana/Roche, Dako/Agilent and Leica Biosystems for p40.
Companion module - run C17
CLDN18.2: This first assessment of CLDN18.2 focused on the identification of patients with gastric and gastroesophageal junction (G/GEJ) cancer to be treated with VYLOY™. 75 laboratories participated and a pass rate of 87% was observed. The CLDN18 (mAb clone 43-14A) RxDx Assays 741-6067 and 740-7037 and corresponding “generic” RTU IHC assays 790-7027 and 744-7162 all from Ventana/Roche were the most successful assays for the evaluation of CLDN18.2 status in gastric carcinomas with pass rates of 100%. LD assays based on concentrated Abs were less successful giving a pass rate of 46%.
PD-L1 TPS/CPS (KEYTRUDA®): 273 laboratories participated being the highest level till now and a pass rate of 76% was observed, being reduced compared to runs C15 and 16. Similar to the observations seen in previous runs, the insufficient PD-L1 IHC results were most frequently characterized by a too low TPS/CPS level changing PD-L1 status in one or more of the carcinomas included. The PD-L1 IHC assay, 22C3 GE006, Dako/Agilent applied in concordance to vendor recommended guidelines, was most successful providing a pass rate of 100%.
July 9th 2025
Søren Nielsen
Scheme director
Run 73 (General Module), B39 (Breast Module), and H27 (HER2 ISH) were completed between January and April 2025. In total, approximately 600 laboratories from more than 50 countries participated in at least one of the offered modules. Around 3.500 slides were assessed overall.
Below is a brief summary of the observed results. Click on the epitope name to access the complete and detailed assessment report for each evaluated marker. Each report outlines best practice methods and recommends immunohistochemical critical assay performance controls (iCAPCs) to support both the setup of IHC protocols and the ongoing monitoring of laboratory performance.
As specified previously, we do encourage participants to update the submitted protocol settings and reagents used if changed from initial entered data. Protocols can be updated until the assessment of the individual run. The submitted data are used to analyze the overall performance and identify root causes for both inferior and optimal performance.
General module - run 73
ALK (lung): 307 laboratories participated and obtained a surprisingly low pass rate of 59% (21% optimal). The Dako/Agilent Ready-To-Use (RTU) system based on the mAb clone OTI1A4 was the most successful assay with a pass rate of 100%. The Ventana RTU system based on the rmAb clone D5F3 was the most used assay for ALK (lung) giving an overall pass rate of 55% being significantly inferior to previous assessment runs.
ASMA: 386 laboratories participated and obtained a pass rate of 73% (39% optimal) being the highest level in all seven assessment runs for ASMA. The mAb clones 1A4, asm-1, rmAb clones EP188 and QR110 could all be used to obtain an optimal staining result. The widely used mAb clone 1A4 was found challenging on the BenchMark IHC platforms. The RTU systems from Dako/Agilent and Leica Biosystems based on mAb clone 1A4 and asm-1, respectively, were most successful and provided high proportions of sufficient and optimal results.
BCL2: 437 laboratories participated and obtained a high pass rate of 93% (70% optimal). The high pass rate was related to the access to and widely used robust Abs as clones 124, BCL-2/100/D5, and SP66. Also, the extensive use of well performing RTU systems from the main IHC system providers impacted the pass rate positively.
GATA3: 422 laboratories participated and obtained a pass rate of 81% (54% optimal) being the highest level for GATA3 to date. The mAb clone L50-823 and the rmAb clones EP368 and QR018 could all be used for an optimal demonstration of GATA3, especially when applied by 3-step detection systems. The Ventana/Roche RTU system based on mAb clone L50-823 was most successful and gave a pass rate of 98% when used with a 3-step detection system and applied otherwise by vendor recommended protocol settings.
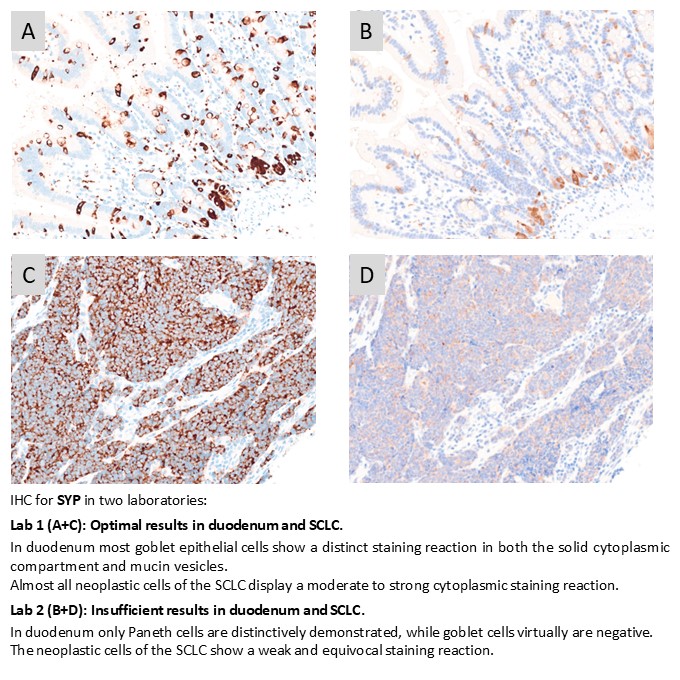
SYP: 429 laboratories participated and obtained a pass rate of 75% (56% optimal). The mAb clone DAK-SYNAP and rmAb clone SP11 were most successful, whereas the widely used clones MRQ-40 and 27G12 provided an inferior performance. The Dako/Agilent RTU system based on mAb clone DAK-SYNAP for Dako Omnis gave an extraordinary performance with a pass rate of 100% and 100% optimal results (n=59).
Breast module - run B39
Estrogen receptor (ER): 430 laboratories participated and obtained a relatively low pass rate of 77% (25% optimal). In this run, the most robust performance was obtained when using the rmAb clone EP1 either as a concentrated format or within an RTU system. Too weak or false negative staining reactions were the predominant features of insufficient results, but also false positive results were observed and related to clone 6F11.
Progesterone receptor (PR): 427 laboratories participated and a pass rate of 98% (77% optimal) was observed. In the latest 5 assessment of PR a high and satisfactory mean pass rate of 93% has been obtained. mAb clones 16, PgR 636, PgR 1294 and rmAb clone 1E2 could all be used to provide optimal results. 90% of the participants used Ready-To-Use (RTU) systems from Ventana/Roche, Dako/Agilent and Leica Biosystems and in total obtained a pass rate of 99% when applying these assays as “plug-and-play”.
HER2 IHC: 418 laboratories participated and obtained a pass rate of 92% (68% optimal). The well-stablished Ventana/Roche HER2 IHC assays PATHWAY®, 790-2991 and VENTANA HER2 (4B5), 790-4493 and the recently launched Ventana RxDx HER2 4B5, 790-7167 were most widely applied and in total used by 71% of all participants giving a pass rate of 96% when used by vendor recommended protocol settings. As also seen in latest assessments, the HercepTest™, GE001, Dako/Agilent, for the Omnis platform was most successful with a pass rate of 100% and 83% optimal results using vendor recommended protocol settings. Many assays were successful for HER2 classical overexpression but showed a decreased agreement and concordance for HER2 Low compared to the level expected and defined by the NordiQC reference methods.
HER2 ISH module - run H27
HER2 ISH: 174 laboratories participated in the technical assessment of HER2 ISH (BRISH) and obtained a pass rate of 85% (45% optimal). The vast majority of participants (97%) used the VENTANA HER2 Dual ISH DNA Probe Cocktail (800-6043). The insufficient results were most frequently characterized by large negative areas in one or more of the breast carcinoma samples compromising the evaluation of HER2/chr 17 ratio, but also caused by impaired morphology, generally weak or missing signals for either HER2 and/or chr17. 217 laboratories participated in the HER2 ISH scoring module and the consensus rate was 90%.
April 30th 2025
Søren Nielsen
Scheme director

For patients with unresectable or metastatic HER2 negative G/GEJ cancer, zolbetuximab (Vyloy™, Astellas) can be offered as first-line treatment providing the tumour is CLDN18.2 positive. In clinical trials as SPOTLIGHT and GLOW, about 38% of patients were categorized as CLDN18.2 positive, defined as ≥75% of tumor cells demonstrating moderate to strong membranous CLDN18 immunohistochemical staining reaction.
The pilot run for CLDN18.2 will be free of charge for participants within the Companion Module run C17. Other participants can contact NordiQC for free admission to this pilot run. Please note that number of participants is limited to 75 and based on “first-come, first-served”. UPDATE: Limit of 75 has been reached.
Run 72 (General module), B38 (breast module), C16 (Companion module) and H26 (HER2 ISH module) were accomplished from October to December 2024. In total, about 550 laboratories from more than 50 countries participated in at least one of the modules offered, and about 3.300 slides were assessed.
Below, find a short summary of the results observed. Click on the epitope name to see the complete and detailed assessment report for the individual markers evaluated. In each assessment report, NordiQC intended to identify best practice methods and recommendable immunohistochemical critical assay performance controls (iCAPCs) to both guide the IHC protocol set-up and to monitor the IHC performance within the laboratories.
As specified previously, we do encourage participants to update the submitted protocol settings and reagents used if changed from initial data of entry. The submitted data are used to analyze the overall performance and identify root causes for both inferior and optimal performance.
Please be aware that we now have opened for protocol submission for the Winter run: General module run 73, Breast module run B39 and HER2 ISH module H27.
At this point we want to notify that in 2025, NordiQC will change the Companion Module for PD-L1 IHC. Only the segment for PD-L1 TPS/CPS will be offered, whereas the segment for PD-L1 IC score will be terminated. The number of participants for PD-L1 TPS/CPS is consistently increasing and in addition more indications are being scored by TPS/CPS.
The change will allow other companion diagnostic assays to be offered by NordiQC and in 2025 we will initiate a pilot-assessment of Claudin 18.2 in gastric cancer. This biomarker is expected to be available in run C17, Spring.
General module - run 72
CD8: 374 laboratories participated and obtained a relatively low pass rate of 74% (55% optimal). The mAb clones C8/144B, 4B11 and the rmAb clone SP239 were all robust antibodies for demonstration of CD8. The two rmAb clones SP16 and SP57 gave an inferior performance with a pass rate of only 1% (1 of 76 protocols) related to aberrant false positive results and the two clones cannot be recommended.
Ki67: 443 laboratories participated and obtained a pass rate of 90% (69% optimal). The assessment focused on Ki67 proliferation index in the classification of neuroendocrine neoplasias. The mAb clones MIB-1 and K2 and the rmAb clones 30.9 and SP6 were all recommendable and robust Abs for Ki67. Overall RTU Abs showed superior pass rates compared to concentrated Abs for Ki67 (92% and 83%, respectively).
MSH6: 379 laboratories participated and obtained a pass rate of 90% (50% optimal) being at the same high level as seen in the latest NordiQC assessment performed. Similar to the previous run, the main parameters contributing to the high pass rate were related to the access to and widely used robust Abs as clone EP49, EPR3945 and BSR100 on the expense of the unsuccessful mAb clone 44. Also, the extensive use of well performing RTU systems from the main IHC system providers impacted the pass rate positively.
p16: 430 laboratories participated and obtained a pass rate of 88% (56% optimal) being the highest level for p16 to date. The assessment focused on p16 in both cervical samples and HNSCC. The positive development was mainly related to the access to newly introduced robust concentrated Abs as clone MX007 and extensive use of well performing RTU systems. In this context both the newly launched p16 RTU system GA783 from Dako/Agilent and the established RTU system 805/825-4713 from Ventana/Roche both gave a high pass rate of 100% and 97%, respectively, when applied by recommended protocol settings.
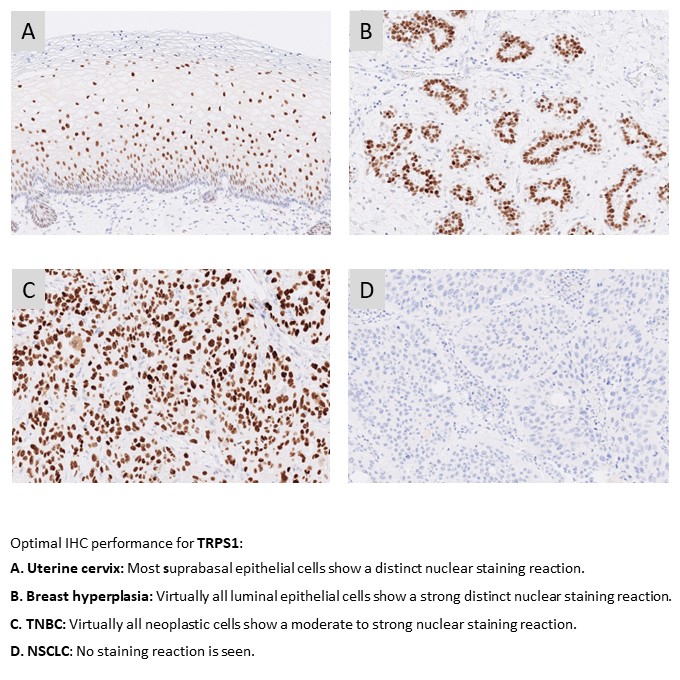
TRPS1: 89 laboratories participated in this first assessment of TRPS1 with a pass rate of 73% (39% optimal). The assessment focused on TRPS1 to identify breast carcinomas including triple negative breast carcinomas in the diagnostic work-up of carcinomas of unknown primary origin. The most cited and in this assessment widely used pAb PA5-84874 from Invitrogen gave a pass rate of 64% and 0% optimal. A low reproducibility was observed as identical protocols based on pAb PA5-84874 gave both sufficient and insufficient results. The rmAb clone ZR382 was found superior providing a pass rate of 94% when used either as concentrated or RTU format.
Breast module - run B38
Estrogen receptor (ER): 438 laboratories participated and obtained a pass rate of 87% (54% optimal). In this run, the most robust performance was obtained when using the two RTU systems of rmAbs clone EP1 for Dako Omnis and clone SP1 for Benchmark Ultra/Ultra PLUS/GX used as true “plug and play” (vendor-recommended protocol settings) assays giving a pass rate of 95%. Too weak or false negative staining reactions were the predominant features of insufficient results, but also false positive results were observed and related to clone 6F11.
HER2 IHC: 418 laboratories participated and obtained a pass rate of 90% (65% optimal). In this assessment, the HercepTest™, GE001, Dako/Agilent, for the Omnis platform was most successful with a pass rate of 100% and 82% optimal results using vendor recommended protocol settings. The newly launched FDA-/CE-IVD approved assay Ventana RxDx HER2 4B5, 790-7167 (Ventana/Roche) was also very successful giving a pass rate of 100%, 71% optimal. Laboratory developed tests based on RTU Abs without predictive claim or based on concentrated Abs gave a pass rate of 75%, 42% optimal. Many assays were successful for HER2 classical overexpression but showed a decreased agreement and concordance for HER2 Low compared to the level expected and defined by the NordiQC reference methods.
Companion module - run C16
PD-L1 TPS/CPS (KEYTRUDA®): 267 laboratories participated and pass rate of 87% (44% optimal) was observed being slightly superior to the mean level of 83% obtained in runs C1-C16. The PD-L1 IHC pharmDx assay, 22C3 GE006, Dako/Agilent applied in concordance to the vendor recommended guidelines. In this assessment the majority of insufficient results were related to technical issues e.g. related to extensive cytoplasmic staining reaction, poor signal-to-noise ratio, etc., observed in one or more of the NSCLCs and TNBC.
PD-L1 IC score (TECENTRIQ®): 141 laboratories participated and a relatively low pass rate of 71% (50% optimal) was observed. The PD-L1 SP142 companion diagnostic IHC assay product no. 741-4860 and the corresponding generic RTU IHC assay 790-4860 both from Ventana/Roche were the most successful assays providing a pass rate of 91% and 85%, respectively. The insufficient results were characterized by either pure false negative results or a false positive IC result of the urothelial carcinoma, tissue core no. 8, and/or an extensive staining reaction in tumour cells compromising the evaluation of PD-L1 reaction in immune cells (non-SP142 based assays).
HER2 ISH module - run H26
HER2 ISH: 175 laboratories participated in the technical assessment of HER2 ISH (BRISH) and obtained a pass rate of 78% (48% optimal). The vast majority of participants (98%) used BRISH HER2 systems from Ventana/Roche. 94% used the VENTANA HER2 Dual ISH DNA Probe Cocktail (800-6043) and 4% the INFORM™ HER2 Dual ISH assay (780-4422/800-4422). The insufficient results were most frequently characterized by large negative areas in one or more of the breast carcinoma samples compromising the evaluation of HER2/chr 17 ratio, but also caused by impaired morphology, generally weak or missing signals for either HER2 and/or chr17.
214 laboratories participated in the HER2 ISH scoring module and the consensus rate was 93%.
December 10th 2024
Søren Nielsen
Scheme director
Run 71 (General module) and C15 (Companion module) were accomplished April to July 2024.
Below, find a short summary of the results observed. Click on the epitope name to see the complete and detailed assessment report for the individual markers evaluated. In each assessment report, NordiQC intended to identify best practice methods and recommendable immunohistochemical critical assay performance controls (iCAPCs) to both guide the IHC protocol set-up and to monitor the IHC performance within the laboratories.
As specified previously, we do encourage participants to update the submitted protocol settings and reagents used if changed from initial data of entry. Protocols can be updated until the slide return deadline. The submitted data are used to analyze the overall performance and identify root causes for both inferior and optimal performance.
General module - run 71
BAP1: 224 laboratories participated in this second assessment and obtained a pass rate of 63% (25% optimal) being slightly reduced compared to the first run 65, 2022. The relatively low pass rate was in particular caused by lack of reproducibility of the IHC assays for BAP1.
CD20: 428 laboratories participated and obtained a relatively high pass rate of 85% (73% optimal). This was the fourth NordiQC assessment of CD20 and the pass rate decreased significantly compared to the previous run 35, 2012. 97% used the mAb clone L26 either as concentrated or RTU format. The RTU systems from Leica Biosystems and Ventana/Roche provided an impressive pass rate of 100% applying vendor recommended protocol settings. The reduced pass rate was especially related to an inferior pass rate of the Dako/Agilent RTU system for Omnis used by 18% of all participants with an overall low pass rate of 42%.
CD117: 428 laboratories participated and obtained a pass rate of 94% (71% optimal) being at the highest level in all runs for CD117. The main parameters contributing to the positive development have in this and the latest runs been related to the extended use of robust primary rmAb clones YR145 and EP10 on the expense of the less successful Abs as pAb 4502 and especially the rmAb clone 9.7. Throughout the different assessment runs a harmonization of the protocol settings has been observed. While previously omission of HIER or HIER in low pH buffers were frequently applied, now virtually all protocols are based on HIER in high pH buffers.
CK-PAN: 410 laboratories participated and obtained a pass rate of 70% (50% optimal). The mAb clone cocktail AE1/AE3 either as “single” cocktail or in combination with other clones was used by 92% of the participants. The performance of the cocktails was highly influenced by choice of pre-treatment method, IHC platform and “AE1/AE3 vendor”.
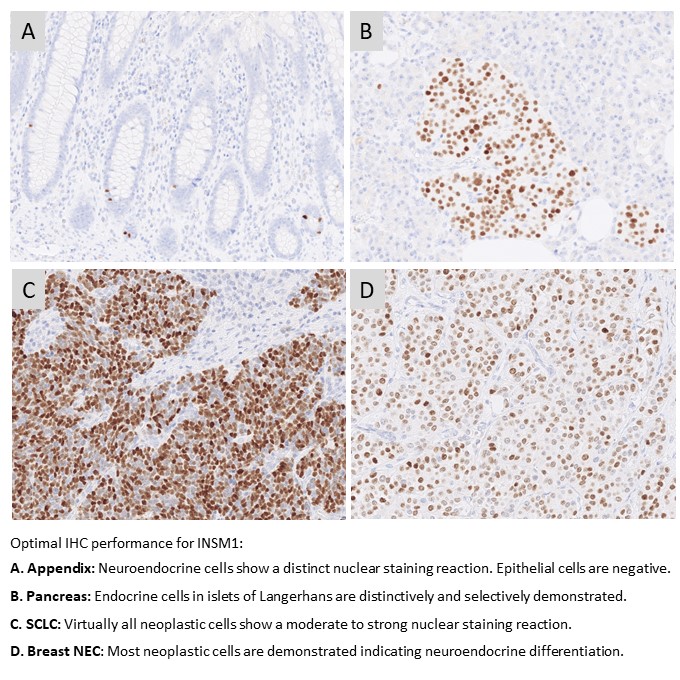
INSM1: 156 laboratories participated in this first assessment of INSM1 with a pass rate of 72% (35% optimal). The assessment focused on INSM1 for the diagnostic work-up in the identification of neuroendocrine differentiation in neoplasias. The most cited and in this assessment widely used mAb clone A-8 gave a pass rate of 55%, when used as a concentrate. A low reproducibility was observed as identical protocols gave both sufficient and insufficient results. The mAb clone MRQ-70 was found superior providing a pass rate of 93% and 94% when used as concentrated or RTU format, respectively.
PMS2: 380 laboratories participated in this fourth assessment of PMS2 and obtained a pass rate of 63% (35% optimal). 85% of the participants used RTU systems for PMS2. The RTU systems from Dako/Agilent and Leica Biosystems based on the rmAb clone EP51 were most successful giving a pass rate of 96% and 100%, respectively applying vendor recommended protocol settings. In contrast an inferior pass rate of 32% was observed for the widely used Ventana/Roche RTU system based on mAb clone A16-4.
Companion module - run C15
PD-L1 TPS/CPS (KEYTRUDA®): 255 laboratories participated being the highest level till now and a very satisfactory pass rate of 88% was observed. Similar to the observations seen in previous runs, the insufficient PD-L1 IHC results were most frequently characterized by a too low TPS/CPS level changing PD-L1 status in one or more of the carcinomas included. The PD-L1 IHC assay, 22C3 GE006, Dako/Agilent applied in concordance to vendor recommended guidelines, was most successful providing a pass rate of 100%, with an optimal rate of 97%. The Ventana/Roche SP263 assay provided a pass rate of 92%, 48% optimal and improved compared to the level in run C14.
PD-L1 IC score (TECENTRIQ®): 146 laboratories participated and a relatively low pass rate of 68% was observed, however slightly increased compared to the level seen in the previous run. The assessment for PD-L1 IC score focused on the evaluation of the analytical accuracy of the PD-L1 IHC assays for urothelial carcinomas and TNBCs. The insufficient results were characterized by either pure false negative results (seen for SP142 based assays), false positive results (seen for non-SP142 based assays) or an extensive staining reaction in tumour cells compromising the evaluation of PD-L1 reaction in immune cell. In concordance with previous runs, the Ventana/Roche PD-L1 SP142 IHC assays 741-4860 and 790-4860 were most successful providing a pass rate of 92% and 95%, respectively.
July 10th 2024
Søren Nielsen
Scheme director
Run 70 (General module), B37 (breast module), and H25 (HER2 ISH module) were accomplished from February to April 2024. In total, about 500 laboratories from more than 50 countries participated in at least one of the modules offered, and about 3.200 slides were assessed.
As usual, a short summary of the results is given below. Click on the epitope name to see the complete and detailed assessment report for the individual markers evaluated. In each assessment report, NordiQC intended to identify best practice methods and recommendable immunohistochemical critical assay performance controls (iCAPCs) to both guide the IHC protocol set-up and to monitor the IHC performance within the laboratories.
As specified previously, we do encourage participants to update the submitted protocol settings and reagents used if changed from initial data of entry. The submitted data are used to analyze the overall performance and identify root causes for both inferior and optimal performance.
Due to the relatively low pass rate, GATA3 and p53 will be repeated in 2025 and reassessment will not be available.
General module - run 70:
Bcl-6: 373 laboratories participated and obtained a relatively low pass rate of 70% (35% optimal). The mAb clones PG-B6p and LN22 provided the highest proportion of sufficient and optimal results applied as an RTU format (Dako/Agilent (Omnis) and Leica Biosystems, respectively). The RTU system based on the mAb clone GI191E/A8 (Ventana/Roche) provided a lower pass rate and was challenged by poor signal-to-noise ratio and false positive results. In a concentrated format, mAb clone GI191E/A8 obtained similar pass rate as the mAb clones PG-B6p and LN22.
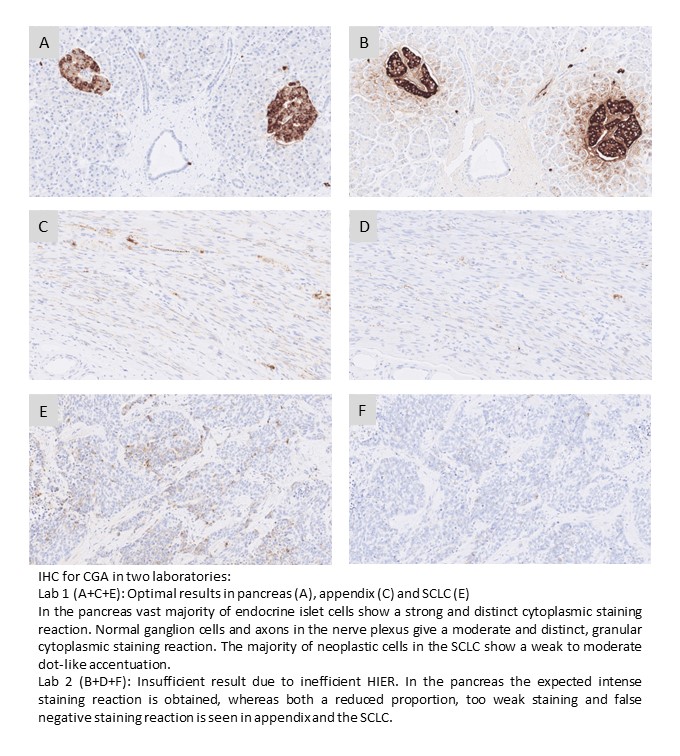
CGA: 369 laboratories participated and obtained a pass rate of 75% (40% optimal). An increased pass rate was observed compared to the previous run 67 and back to the level seen in run 53. The mAb clone LK2H10 was most widely used and very successful and in particular as RTU from Ventana/Roche. In concordance with previous assessments for CGA, the mAb clones DAK-A3 and 5H7 were found less successful and cannot be recommended.
GATA3: 390 laboratories participated and obtained a low pass rate of 65% (40% optimal). This was the fourth assessment of GATA3 in NordiQC and focused on the application in the diagnostic work-up of cancers of unknown primary origin. The vast majority of participants used the mAb clone L50-823. Used within a LD assay, optimal results could be obtained on all main fully automated IHC platforms. The Ventana/Roche RTU system based on clone L50-823 gave the highest pass rate and proportion of optimal results. Due to the relatively low pass rate, GATA3 will be repeated in 2025 and reassessment will not be available.
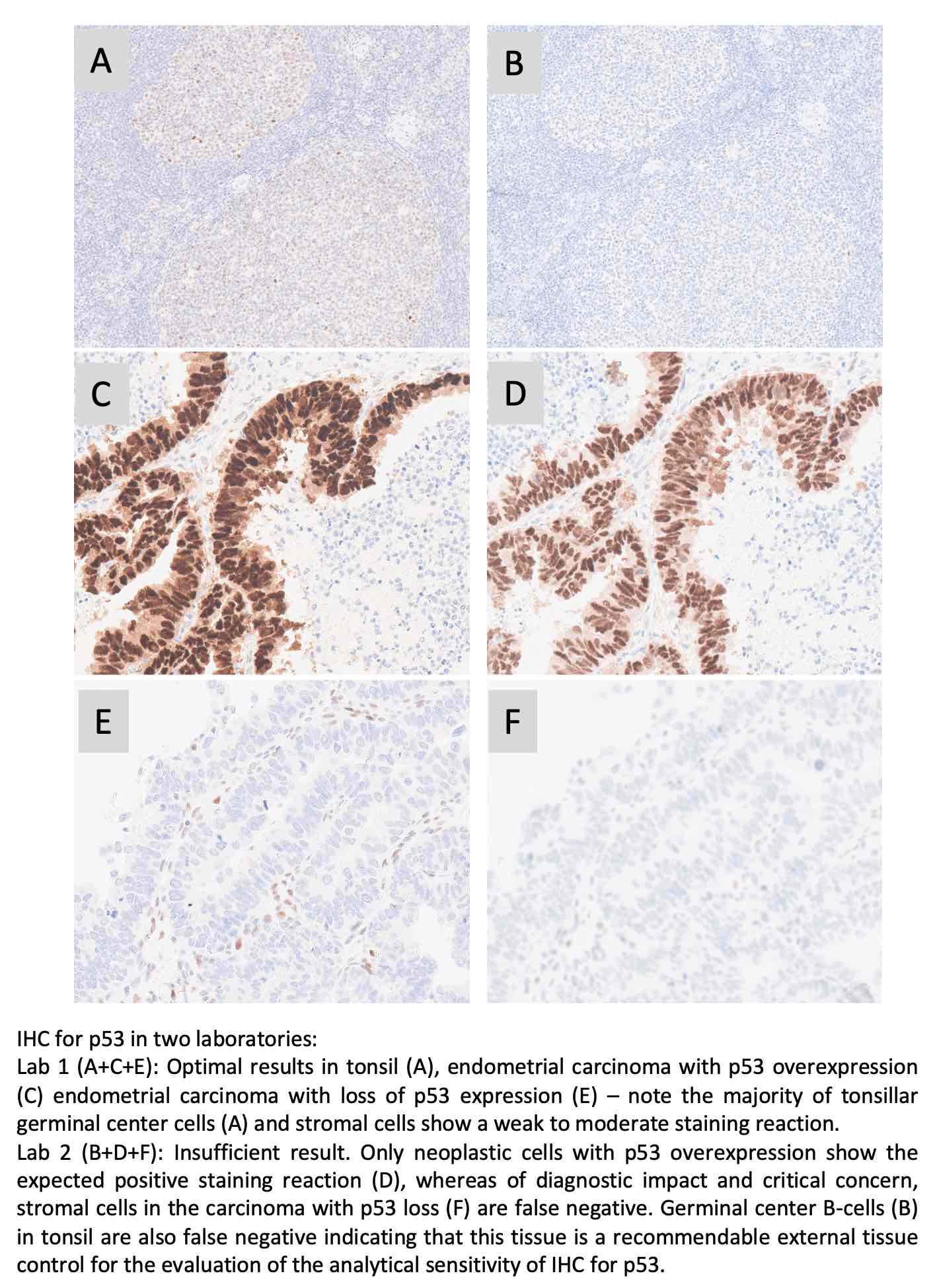
p53: 396 laboratories participated and obtained a pass rate of 65% (16% optimal). This was the sixth assessment of p53 in NordiQC, and the fourth using more challenging assessment criteria introduced in run 63, 2021, focusing on IHC for p53 to demonstrate different TP53 mutations in endometrial carcinomas. The pass rate was identical to the latest run 67 being 65% (29% optimal). The widely used mAbs clones BP53-11 and DO-7 could both be used to provide optimal results providing the protocols were based on efficient HIER in an alkaline buffer and of central importance in combination with a 3-step detection system. As a consequence of the “new purpose of p53 IHC” the most important parameter being a careful calibration of the primary Ab to consistently identify p53 in low-level expressing structures as dispersed normal lymphocytes and stromal cells being of central importance in the diagnosis of carcinomas with loss of p53 expression. Due to the relatively low pass rate, p53 will be repeated in 2025 and reassessment will not be available.
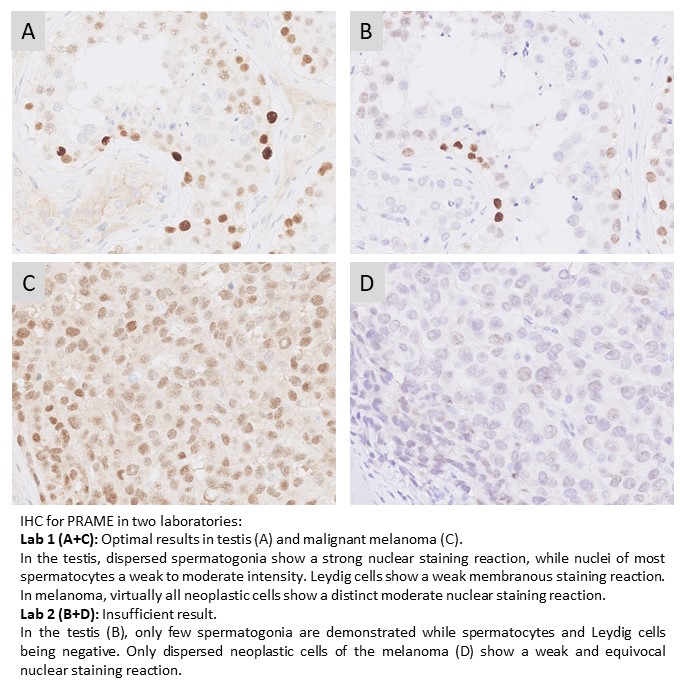
PRAME: 259 laboratories participated and a pass rate of 78% was observed (44% optimal). This was the second assessment for PRAME in NordiQC, and a slightly improved pass rate was obtained, compared to 73% (38% optimal) in the last run. The assessment focused again on PRAME being used in the diagnostic work-up in the differential diagnosis between malignant melanoma and benign melanocytic lesions. The most widely used rmAb clone EPR20330 proved to be most robust with a pass rate of 80%, 44% optimal and provided optimal results on all the main automated IHC platforms (Dako/Agilent, Leica Biosystems and Ventana/Roche). Both false negative and false positive results were observed.
Breast module - run B37:
Estrogen receptor (ER): 426 laboratories participated and obtained a pass rate of 85% (35% optimal) being increased to the latest assessment, but still slightly reduced to the level seen previously. In concordance to previously, too weak or false negative staining reactions were the predominant features of insufficient results. The widely used rmAb clones SP1 and EP1 and mAb clone 6F11 could all be used to provide an optimal result. 91% of the participants used RTU systems for the demonstration of ER. The RTU system from Dako/Agilent based on clone EP1 for Omnis was most successful providing a pass rate of 96%. Tonsil was found to be recommendable as positive and negative tissue control.
HER2 IHC: 405 laboratories participated and obtained a pass rate of 89% (57% optimal) being slightly increased compared to run B36 and similar to the level observed in runs B35. In this assessment, the recently launched 2’ generation HercepTest™ Dako/Agilent for Omnis was most successful providing a pass rate of 100%. The well-established CE-IVD approved HER2 IHC assays PATHWAY® and HER2 4B5, Ventana/Roche gave a slightly lower pass rate of 91% and 87% respectively, compared to 95% and 94% in run B36 when applied as recommended by vendor. LD assays based on a concentrate or RTU with no predictive claim were used by 18% of participants giving a pass rate of 73% (31% optimal). The insufficient results were primarily caused by false negative results, whereas a minor proportion caused by either an increased proportion of positive cells or an excessive cytoplasmic staining reaction compromising the read-out.
Progesterone receptor (PR): 420 laboratories participated and a pass rate of 93% (71% optimal) was observed being virtual identical to the level seen in the last four runs. mAb clones 16, PgR 636, PgR 1294 and rmAb clone 1E2 could all be used to provide optimal results. 88% of the participants used RTU systems and obtained a pass rate of 95%. In this assessment, too weak or false negative staining reactions were the predominant features of insufficient results. Tonsil and uterine cervix in combination are recommendable negative and positive tissue controls.
HER2 ISH module - H25:
HER2 ISH: 161 laboratories participated in the technical assessment of HER2 ISH (BRISH) and obtained a pass rate of 85% (45% optimal) and being the highest pass rate seen in the HER2 ISH in the last decade. The improvement was primarily related to the changed assessment criteria from run H24, allowing large negative areas of >25% providing the individual tissue cores still could be evaluated with confidence. The VENTANA HER2 Dual ISH DNA Probe Cocktail, 800-6043 was used by 94% of the participants either as single assay or in combination with HER2 IHC. The insufficient results were typically characterized by large negative areas in one or more of the breast carcinoma samples compromising the evaluation of HER2 gene status, but also caused by impaired morphology, generally weak or missing signals.
213 laboratories participated in the HER2 ISH scoring module and the consensus rate was 91%.
April 28th 2024
Søren Nielsen
Scheme director
Run 69 (General module), B36 (breast module), C14 (Companion module) and H24 were accomplished from October to December 2023. In total, about 500 laboratories from more than 40 countries participated in at least one of the modules offered, and about 3.200 slides were assessed.
As usual, a short summary of the results is given below. Click on the epitope name to see the complete and detailed assessment report for the individual markers evaluated. In each assessment report, NordiQC intended to identify best practice methods and recommendable immunohistochemical critical assay performance controls (iCAPCs) to both guide the IHC protocol set-up and to monitor the IHC performance within the laboratories.
As specified previously, we do encourage participants to update the submitted protocol settings and reagents used if changed from initial data of entry. The submitted data are used to analyze the overall performance and identify root causes for both inferior and optimal performance.
Please be aware that we now have opened for protocol submission for the Winter run: General module run 70, Breast module run B37 and HER2 ISH module H25.
General module - run 69
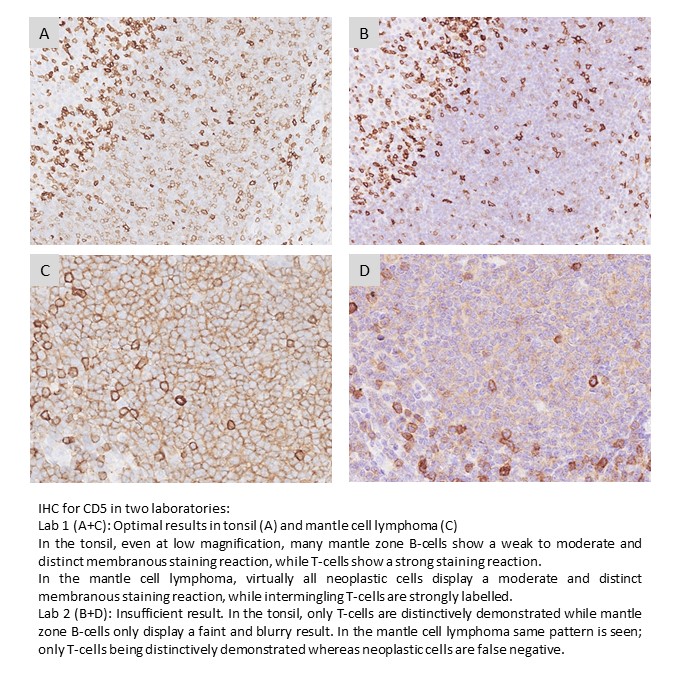
CD5: 379 laboratories participated and obtained a relatively low pass rate of 72% (54% optimal). Several antibody clones could be used to obtain an optimal result. The rmAb clone SP19 proved to be the most robust antibody for the detection of CD5 achieving a pass rate of 95%?(177/187) with 81% optimal across all formats and products based on that clone. Protocols based on the mAb clone 4C7 was challenged by low analytical sensitivity and 50% of all insufficient results were caused by less successful performance of this clone either as concentrated format or RTU system from Dako/Agilent.
CD138: 386 laboratories participated and obtained a pass rate of 86% (60% optimal). The two widely used mAb clones MI15 and B-A38 provided optimal results on all the main fully automated IHC platforms both as concentrated format and corresponding RTU systems. A 100% pass rate was observed for the Leica Biosystems and Dako/Agilent RTU systems based on the mAb clone MI15 when applied in compliance with vendor recommended protocol settings.
CK8/18: 282 laboratories participated and obtained a pass rate of 85% (65% optimal) being at the highest level in the nine NordiQC assessments performed. The extended use of highly sensitive and robust antibodies as the clone cocktails B22.1/B23.1 and EP17/30 on the expense of the less successful clone DC10 has been a main contributor for the improved pass rate. In addition, the identification of liver as a reliable critical tissue control to evaluate a successful IHC assay for CK8/18 in the diagnostic work-up for CUP has been instrumental for the improvement.
EpCAM: A record high number of 345 laboratories participated and obtained a pass rate of 48% (15% optimal). As seen in previous assessments for EpCAM, the selection of antibody must be made with focus on the IHC platform and reagents available in the laboratory. The most commonly used mAb clone Ber-EP4 requires HIER in a special formulated buffer available for the Dako/Agilent IHC platforms and here provides high pass rates both within LD assays or as RTU system for Dako Omnis. On e.g. the Ventana BenchMark IHC platforms, mAb clones BS14 and VU-1D9 had significantly higher pass rates compared to mAb clones Ber-EP4 and MOC-31.
PSA: 377 laboratories participated and a pass rate of 85% was observed (50% optimal). As concentrated format within a laboratory developed test, mAb clone 35H9 and rmAb EP109 were most successful, whereas the mAb clone ER-PR8 proved to be more challenging and needed a carefully calibration of both antibody titer and choice of detection system. The RTU systems from Ventana/Roche and Leica Biosystems based on a pAb and mAb clone 35H9, respectively were very successful providing a high proportion of sufficient and optimal results.
Breast module - run B36
Estrogen receptor (ER): 440 laboratories participated and obtained a pass rate of 78% being reduced compared to the levels obtained in the latest assessments. Too weak or false negative staining reactions were the predominant features of insufficient results. The widely used rmAb clones SP1 and EP1 and mAb clone 6F11 could all be used to provide an optimal result. 91% of the participants used RTU systems for the demonstration of ER. The RTU system from Dako/Agilent based on clone EP1 for Omnis was most successful providing a pass rate of 91%. Tonsil was found to be recommendable as positive and negative tissue control.
HER2 IHC: 419 laboratories participated and obtained a pass rate of 86% (53% optimal) being slightly reduced compared to run B35 but on par to the level observed in runs B33 and B34. In this assessment, the well-established CE-IVD approved HER2 IHC assays PATHWAY® and HER2 4B5, Ventana/Roche, but also the recently launched 2’ generation HercepTest™ Dako/Agilent for Omnis were most successful providing a pass rate of 93%-94%. The “classical” Dako/Agilent HercepTest™ for Autostainer gave a disappointing pass rate of 38%. LD assays based on a concentrate or RTU with no predictive claim were used by 20% of participants giving a pass rate of 72% (28% optimal). The insufficient results were primarily caused by false negative results, whereas a minor proportion caused by poor signal-to-noise ratio compromising the read-out or false positive results.
Companion module - run C14
PD-L1 TPS/CPS (KEYTRUDA®): 243 laboratories participated and an unexpected and relatively low pass rate of 69% was obtained being significantly reduced compared to the latest runs. The PD-L1 IHC pharmDx assay, 22C3 GE006, Dako/Agilent applied in concordance to the vendor recommended guidelines, was most successful providing a pass rate of 100%, 90% optimal. Surprisingly and as yet unexplained due to unknown reasons, the Ventana/Roche PD-L1 IHC assays 741-4905 and 740-4907 for BenchMark (Ultra Plus/Ultra/XT/GX) based on the rmAb clone SP263 provided an overall low pass rate of 40% being inferior to the level expected and obtained in previous runs. LD assays based on concentrated formats and RTU systems without predictive claims provided a pass rate of 76%.
PD-L1 IC score (TECENTRIQ®): 129 laboratories participated and a low pass rate of 56% was observed being reduced from the level of 68% obtained in run C13. The two PD-L1 SP142 CDx IHC assays product no. 741-4860 and 790-4860, Ventana/Roche were as seen previously the most successful assays providing a pass rate of 66% and 67%, respectively but reduced from recent levels. PD-L1 CDx assays as SP263 (741-4905, Ventana/Roche) and 22C3 (SK006/GE006, Dako/Agilent) being successful in the PD-L1 TPS/CPS assessments provided only few sufficient results. Insufficient results were characterized by either pure false negative results (seen for SP142) or false positive results often combined with extensive reaction in tumour cells in the carcinomas compromising the scoring of PD-L1 in immune cells (non-SP142 based assays).
HER2 ISH module - run H24
HER2 ISH: 176 laboratories participated in the technical assessment of HER2 ISH (BRISH) and obtained a pass rate of 79% (36% optimal). The improvement was primarily related to the changed assessment criteria, allowing large negative areas of >25% providing the individual tissue cores still could be evaluated with confidence. The VENTANA HER2 Dual ISH DNA Probe Cocktail, 800-6043 was used by 89% of the participants either as single assay or in combination with HER2 IHC. The insufficient results were typically characterized by large negative areas in one or more of the breast carcinoma samples compromising the evaluation of HER2 gene status, but also caused by impaired morphology, generally weak or missing signals.
225 laboratories participated in the HER2 ISH scoring module and the consensus rate was 94%, and 76% for laboratories using FISH and BRISH, respectively.
December 11th 2023
Søren Nielsen
Scheme director
Run 68 (General module) and C13 (Companion module) were accomplished April to July 2023.
As usual, a short summary of the results is given below. Click on the epitope name to see the complete and detailed assessment report for the individual markers evaluated. In each assessment report, NordiQC intended to identify best practice methods and recommendable immunohistochemical critical assay performance controls (iCAPCs) to both guide the IHC protocol set-up and to monitor the IHC performance within the laboratories.
As specified previously, we do encourage participants to update the submitted protocol settings and reagents used if changed from initial data of entry. Protocols can be updated until the slide return deadline. The submitted data are used to analyze the overall performance and identify root causes for both inferior and optimal performance.
Due to the relatively low pass rate and being a new IHC test, PRAME will be repeated in 2024 and reassessment will not be available.
General module - run 68
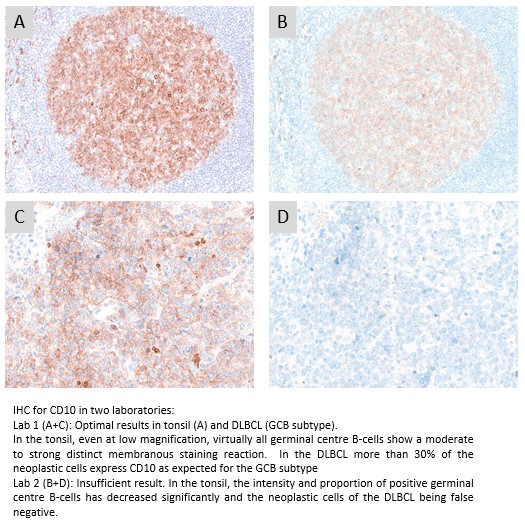
CD10: 403 laboratories participated and obtained a pass rate of 73% (48% optimal) being a slight improvement compared to the recent run 66. The widely used mAb clone 56C6 provided an optimal result on all the main fully automated IHC platforms both as concentrated format and corresponding RTU systems. The recently launched RTU system from Dako/Agilent based on mAb clone DAK-CD10 for Dako Omnis was most successful both when used by vendor recommended or modified protocol settings. As seen in run 66, the Ventana/Roche RTU system based on rmAb clone SP67 being applied by 33% of all participants showed an inferior performance.
MSH2: 350 laboratories participated and obtained a relatively high pass rate of 91% (62% optimal). This was the fifth NordiQC assessment of MSH2 and a consistent improved performance has been observed since 2014. The extended use and access to high quality IHC RTU systems from the main IHC vendors have impacted positively to the pass rate. In total 88% of the participants used RTU systems. Especially the RTU systems for fully automated IHC systems from the three main providers (Dako/Agilent, Leica Biosystems and Ventana/Roche) were successful, providing a pass rate of 100%, compared to 73% for laboratory developed assays based on concentrates.
PAX8: 368 laboratories participated and obtained a pass rate of 54% (32% optimal). The pass rate has now improved slightly but at a slow pace from the low level of 37% in run 56, 2019. In concordance to the previous assessments especially the rmAbs clone SP348 but also QR016 were most successful. In contrast, the widely used mAb clone MRQ-50 provided a poor performance especially on the Ventana BenchMark and Dako Omnis platforms giving a too low analytical sensitivity and at the same time it also gave a cross-reaction to other PAX subtypes as PAX5 in B-cells. The contrast in performance for SP348 versus MRQ-50 was in this assessment quite remarkable; irrespective of protocol settings applied, SP348 gave a pass rate of 91% compared to 50% for MRQ-50.
PRAME: 222 laboratories participated in this first assessment of PRAME and obtained a pass rate of 73% (38% optimal). The assessment focused on PRAME being used in the diagnostic work-up in the differential diagnosis between malignant melanoma and benign melanocytic lesions. The most cited and in this assessment widely used rmAb clone EPR20330 proved to be most robust with a pass rate of 80%, 47% optimal and provided optimal results on all the main automated IHC platforms (Dako/Agilent, Leica Biosystems and Ventana/Roche). Both false negative and false positive results were observed. Due to the relatively low pass rate and being a new IHC test, PRAME will be repeated in 2024 and reassessment will not be available.
TTF1: 401 laboratories participated and obtained a pass rate of 81% (71% optimal) being the all-time highest level in the eight assessment runs performed by NordiQC. In concordance with previous NordiQC assessments for TTF1, the mAb clone SPT24 and the rmAb clone SP141 were most reliable for the demonstration of TTF1 providing pass rates of 97% and 96%, respectively. In contrast, mAb clone 8G7G3/1 was less successful and provided a significantly lower pass rate of 6%. Too weak or false negative staining reactions were seen in 87% of the insufficient results.
URO II/III: 107 laboratories participated in this second assessment of URO II/III and obtained a pass rate of 49% (29% optimal). As observed in the first run, antibodies targeted for URO II were most successful. The concentrated format of mAb clone BC21, within a laboratory developed assay, provided the highest proportion of optimal results, 41%, being observed on all main fully automated IHC platforms.
Companion module - run C13
PD-L1 TPS/CPS: 243 laboratories participated being the highest level till now and a very satisfactory pass rate of 92% was observed. This NordiQC assessment for PD-L1 focused on TPS and CPS status in NSCLC and TNBC, respectively. Similar to observations seen in previous runs, the insufficient PD-L1 IHC results were most frequently characterized by a too low TPS/CPS level changing PD-L1 status in one or more of the carcinomas included. The PD-L1 IHC pharmDx assay, 22C3 GE006, Dako/Agilent applied in concordance to the vendor recommended guidelines, was most successful providing a pass rate of 100%, with an optimal rate of 79%.
PD-L1 IC score: 139 laboratories participated and a relatively low pass rate of 68% was observed, however slightly increased compared to the levels seen in the latest runs. The assessment for PD-L1 IC score focused on the evaluation of the analytical accuracy of the PD-L1 IHC assays for urothelial carcinomas and TNBCs. The insufficient results were characterized by either pure false negative results (seen for SP142 based assays), false positive results (seen for non-SP142 based assays) or an extensive staining reaction in tumour cells compromising the evaluation of PD-L1 reaction in immune cell. In concordance with previous runs, the Ventana/Roche PD-L1 SP142 IHC assay 741-4860 was most successful providing a pass rate of 82%.
July 10th 2023
Søren Nielsen
Scheme director
Run 67 (General module), B35 (breast module) and H23 were accomplished from January to April 2023. In total, about 500 laboratories participated in at least one of the modules offered, and more than 3.200 slides were assessed.
As usual, a short summary of the results is given below. Click on the epitope name to see the complete and detailed assessment report for the individual markers evaluated. In each assessment report, NordiQC intended to identify best practice methods and recommendable immunohistochemical critical assay performance controls (iCAPCs) to both guide the IHC protocol set-up and to monitor the IHC performance within the laboratories.
As specified previously, we do encourage participants to update the submitted protocol settings and reagents used if changed from initial data of entry. The submitted data are used to analyze the overall performance and identify root causes for both inferior and optimal performance.
Due to the relatively low pass rates for CGA and p53, these will be repeated in 2024 and reassessment will not be available.
General module - run 67
CD4: 332 laboratories participated and obtained a pass rate of 80% (61% optimal). The rmAb clone SP35 was most successful providing an optimal result on all the main fully automated IHC platforms both as concentrated format and especially as corresponding RTU system from Ventana/Roche with a pass rate of 98%. The mAb clone 4B12 was widely used both as concentrate and within RTU systems but showed inferior performance on fully automated platforms especially on Dako Omnis. The Dako/Agilent RTU format IS/IR649 for Autostainer based on clone 4B12 was frequently migrated to Dako Omnis (n=28) with an unacceptable pass rate of 0%.
CGA: 365 laboratories participated and obtained a relatively low pass rate of 64% (32% optimal). This was the eigth NordiQC assessment of CGA. A reduced pass rate was observed compared to the previous runs, which mainly was due to less robust Abs as mAb clones DAK-A3 and 5H7 being used by 16% of all participants with a low pass rate of 5%, none optimal. In contrary mAb clone LK2H10 was very successful and in particular as RTU from Ventana/Roche. In concordance with previous assessments for CGA, appendix was found recommendable as positive and negative tissue control. Due to the relatively low pass rate, CGA will be repeated in 2024, run 68 and reassessment will not be available.
MLH1: 342 laboratories participated and obtained a pass rate of 71% (46% optimal). Several Abs could be used for an optimal result. Irrespective of the Ab applied, sufficient HIER, use of a highly sensitive detection system and careful calibration of the primary antibody were the most important prerequisites for an optimal staining result. Overall, the mAb clone ES05 (both concentrate and as RTU) applied on the Leica Bond stainer platforms provided the highest pass rate of 94%, 82% optimal when used with an alkaline HIER buffer and a 3-step detection system.
p40: 343 laboratories participated and obtained a pass rate of 85% (60% optimal). The assessment focused on p40 being used in the diagnostic work-up in in the differential diagnosis between lung squamous cell carcinoma and lung adenocarcinoma. This was the fourth assessment of p40 in NordiQC and increased and stabile pass rate has been obtained compared to the initial runs. The extended use of the highly sensitive and robust mAb clone BC28 both as concentrate and as RTU format on the expense of pAbs has been instrumental for the improvement. Additionally, the recently launched RTU system from Dako/Agilent based on mAb clone DAK-p40 giving a pass rate of 100% impacted positively on the overall pass rate.
p53: 372 laboratories participated and obtained a pass rate of 65% (29% optimal) being an improvement from 44% and 55% in run 63 and 55, respectively but still significantly inferior the level of 79% in run 38, 2013. Similar to the two previous runs, this assessment focused on IHC for p53 to demonstrate different TP53 mutations in endometrial carcinomas. The widely used mAbs clones BP53-11 and DO-7 could both be used to provide optimal results providing the protocols were based on efficient HIER in an alkaline buffer and of central importance in combination with a 3-step detection system. As a consequence of the “new purpose of p53 IHC” the most important parameter being a careful calibration of the primary Ab to consistently identify p53 in low-level expressing structures as dispersed normal lymphocytes and stromal cells being of central importance in the diagnosis of carcinomas with loss of p53 expression. Due to the relatively low pass rate, p53 will be repeated in 2024 and reassessment will not be available.
Breast module - run B35
Estrogen receptor (ER): 422 laboratories participated and obtained a pass rate of 91% (58% optimal). The widely used rmAb clones SP1 and EP1 and mAb clone 6F11 could all be used to provide an optimal result. 89% of the participants used RTU systems for the demonstration of ER. The RTU systems from Ventana/Roche based on clone SP1 for BenchMark and Dako/Agilent based on EP1 for Omnis were most successful providing a pass rate of 100% and 98%, respectively. Too weak or false negative staining reactions were the predominant features of insufficient results, but in a few cases false positive results were observed (mAb clone 6F11). Tonsil was found to be recommendable as positive and negative tissue control.
Progesterone receptor (PR): 414 laboratories participated and obtained a pass rate of 92% (60% optimal) being virtual identical to the level seen in the last four runs. mAb clones 16, PgR 636, PgR 1294 and rmAb clone 1E2 could all be used to provide an optimal result. 84% of the participants used RTU systems from Ventana/Roche, Dako/Agilent and Leica Biosystems and in total obtained a pass rate of 95% when applying these assays as “plug-and-play”. The Leica Biosystems’ RTU system based on clone 16 was most successful with 100% optimal results. In this assessment, too weak or false negative staining reactions were the predominant features of insufficient results. Tonsil and uterine cervix in combination are recommendable negative and positive tissue controls.
HER2 IHC: 403 laboratories participated and obtained a pass rate of 90% (66% optimal). In this assessment, the CE-IVD approved HER2 IHC assay PATHWAY®, Ventana/Roche, VENTANA HER2 4B5 and the recently launched HercepTest™ Dako/Agilent for Omnis were most successful providing a pass rate of 95%, 94% and 100%, respectively. LD assays based on a concentrate or RTU with no predictive claim were used by 20% of participants giving an overall pass rate of 79%. The insufficient results were primarily characterized by reduced proportion of positive cells, a too weak or false negative staining reaction
HER2 ISH module - run H23
HER2 ISH: 163 laboratories participated in the technical assessment of HER2 ISH (BRISH) and obtained a pass rate of 59% (37% optimal) and slightly reduced compared to run H22. The VENTANA HER2 Dual ISH DNA Probe Cocktail, 800-6043 was used by 90% of the participants either as single assay or in combination with HER2 IHC. As observed in virtually all latest assessments, the insufficient results were mainly caused by large negative areas and missing signals in >25% of the neoplastic cells in one or more of the samples included.
207 laboratories participated in the HER2 ISH scoring module (65 via FISH and 142 via BRISH) and the consensus rate was 98%, and 80% for laboratories using FISH and BRISH, respectively.
April 26th 2023
Søren Nielsen
Scheme director
Run 66 (General module), B34 (Breast module), C12 (Companion module) and H22 (HER2 ISH module) were accomplished from October to December 2022. In total, about 500 laboratories participated in at least one of the modules offered, and more than 2.900 slides were assessed.
As usual, a short summary of the results is given below. Click on the epitope name to see the complete and detailed assessment report for the individual markers evaluated. In each assessment report, NordiQC intended to identify best practice methods and recommendable immunohistochemical critical assay performance controls (iCAPCs) to both guide the IHC protocol set-up and to monitor the IHC performance within the laboratories.
As specified previously, we do encourage participants to update the submitted protocol settings and reagents used if changed from initial data of entry. The submitted data are used to analyze the overall performance and identify root causes for both inferior and optimal performance.
Please be aware that we now have opened for protocol submission for the Winter run: General module run 67, Breast module run B35 and HER2 ISH module H23.
General module - run 66
BSAP: 259 laboratories participated and obtained a pass rate of 86% (52% optimal). Several antibody clones could be used to obtain an optimal result. The mAb clone DAK-Pax5 was found very robust and the RTU systems from Dako/Agilent based on this clone (IR650 and GA650) provided superior results, when applied by the vendor recommended settings. Protocols based on the mAb clones 1EW, 24 and the rmAb clone SP34 were often challenged by low analytical sensitivity and/or poor signal-to-noise ratio hindering interpretation of the specific signal for BSAP.
CD10: 393 laboratories participated and obtained a relatively low pass rate of 64% (37% optimal). The widely used mAb clone 56C6 provided an optimal result on all the main fully automated IHC platforms both as concentrated format and corresponding RTU systems. The Leica Biosystem RTU system based on mAb clone 56C6 gave a pass rate of 100%, when used by recommended protocol settings. The recently launched RTU system from Dako/Agilent based on mAb clone DAK-CD10 was also very successful, whereas the Ventana/Roche RTU system using rmAb clone SP67 being applied by 32% of all participants showed an inferior performance. Due to the relatively low pass rate, CD10 will be repeated in 2023, run 68 and reassessment will not be available.
Napsin A: 321 laboratories participated and obtained a pass rate of 83% (46% optimal). The pass-rate has improved consistently in the three assessments conducted in NordiQC and mainly related to the extended use of robust and specific monoclonal primary Abs as the clones IP64 and MRQ-60 on the expense on polyclonal Abs typically giving aberrant and false positive results.
SMH: 152 laboratories participated and obtained a pass rate of 81% (58% optimal). The assessment focused on SMH being used in the diagnostic work-up in breast samples to distinguish benign and precursor lesions from invasive cancer. The widely used mAb clone SMMS-1 provided an optimal result on all the main fully automated IHC platforms both as concentrated format and corresponding RTU systems. The Dako/Agilent RTU format IS/IR066 for Autostainer based on clone SMMS-1 was frequently migrated to Dako Omnis with an inferior result giving an unacceptable pass rate of 15%.
SYP: 390 laboratories participated and a pass rate of 70% was observed (47% optimal). This was the seventh NordiQC assessment of SYP. A reduced pass rate was observed compared to the two previous runs, which primarily was due to extended use of RTU systems with less successful vendor recommended protocol settings, off-label use of RTU formats and less robust Abs in general. Duodenum seemed to be a recommendable positive tissue control for SYP using goblet cells as iCAPC for analytical sensitivity.
Breast module - run B34
Estrogen receptor (ER): 406 laboratories participated and obtained a pass rate of 93% (63% optimal). The widely used rmAb clones SP1 and EP1 and mAb clone 6F11 could all be used to provide an optimal result. 89% of the participants used RTU systems for the demonstration of ER. The RTU systems from Ventana/Roche based on clone SP1 for BenchMark and Dako/Agilent based on EP1 for Omnis were most successful providing a pass rate of 94% and 97%, respectively. Too weak or false negative staining reactions were the predominant features of insufficient results. Tonsil was found to be recommendable as positive and negative tissue control.
HER2 IHC: 392 laboratories participated and obtained a pass rate of 84% (70% optimal). In this assessment, the CE-IVD approved HER2 IHC assay PATHWAY®, Ventana/Roche and the recently launched HercepTest™ Dako/Agilent for Omnis were most successful providing a pass rate of 93% and 100%, respectively. The “classical” Dako/Agilent HercepTest™ for Autostainer gave a disappointing pass rate of 33%. LD assays based on a concentrate or RTU with no predictive claim were used by 21% of participants giving a pass rate of 77% (56% optimal). The insufficient results were primarily caused by false negative results or poor signal-to-noise ratio compromising the read-out.
Companion module - run C12
PD-L1 TPS/CPS (KEYTRUDA®): 232 laboratories participated and a pass rate of 85% was observed. This was the second NordiQC assessment of PD-L1 for TPS/CPS status with focus on TNBCs in addition to NSCLCs. Similar to observations seen in previous runs, the insufficient PD-L1 IHC results were most frequently characterized by a too low TPS/CPS level changing PD-L1 status in one or more of the carcinomas included. The PD-L1 IHC pharmDx assay, 22C3 GE006, Dako/Agilent applied in concordance to the vendor recommended guidelines, was most successful providing a pass rate of 100%, with an optimal rate of 79%, being superior to the other companion diagnostic assays and LD assays based on concentrated Abs or RTU systems without predictive claim.
PD-L1 IC score (TECENTRIQ®): 134 laboratories participated and a relatively low pass rate of 64% was observed – slightly increased compared to the level seen in run C11. The assessment for PD-L1 IC score focused on the evaluation of the analytical accuracy of the PD-L1 IHC assays for urothelial carcinomas and TNBCs. The insufficient results were characterized by either pure false negative results (seen for SP142 based assays) or an extensive staining reaction in tumour compromising the evaluation of PD-L1 reaction in immune cells in combination with an either false negative or false positive result (seen for non-SP142 based assays). In concordance with previous runs, the Ventana/Roche PD-L1 SP142 IHC assay 741-4860 was most successful providing a pass rate of 87%.
HER2 ISH module - run H22
HER2 ISH: 165 laboratories participated in the technical assessment of HER2 ISH (BRISH) and obtained a pass rate of 64% (32% optimal) and slightly improved compared to run H21. The VENTANA HER2 Dual ISH DNA Probe Cocktail, 800-6043 was used by 80% of the participants giving a pass rate of 68%. As observed in virtually all latest assessments, the insufficient results were mainly caused by large negative areas and missing signals in >25% of the neoplastic cells in one or more of the samples included.
207 laboratories participated in the HER2 ISH scoring module (59 via FISH and 148 via BRISH) and the consensus rate was 90%, and 66% for laboratories using FISH and BRISH, respectively.
December 16th 2022
Søren Nielsen
Scheme director

NordiQC has gained accreditation status by DANAK for proficiency testing.
The general, breast cancer and companion modules are accredited per 28.11.2022 by DANAK under registration number 616 to proficiency testing according to ISO 17043. NordiQC is affiliated to and based at Department of Pathology, Aalborg University Hospital, Denmark and in 2020 the department was accredited according to ISO 15189 (Medical laboratories – Requirements for quality and competence).
Run 65 (General module) and C11 (Companion module) were accomplished from March to July 2022. In total, 429 laboratories participated in one or both of the modules offered, and more than 2.200 slides and 12.000 individual tissue cores were assessed.
As usual, a short summary of the results is given below. Click on the epitope name to see the complete and detailed assessment report for the individual markers evaluated. In each assessment report, NordiQC intended to identify best practice methods and recommendable immunohistochemical critical assay performance controls (iCAPCs) to both guide the IHC protocol set-up and to monitor the IHC performance within the laboratories.
As specified previously, we do encourage participants to update the submitted protocol settings and reagents used if changed from initial data of entry. The submitted data are used to analyze the overall performance and identify root causes for both inferior and optimal performance.
Please be aware that we now have opened for protocol submission for the Autumn run: General module run 66, Breast module run B34, Companion module run C12 and HER2 ISH module H22.
General module - run 65
ALK (lung): 256 laboratories participated and obtained a pass rate of 77% (59% optimal). The mAb clones 5A4, OTI1A4 and rmAb clone D5F3 could all both as concentrated format in a LD assay and within a RTU system provide an optimal result. The recently launched RTU system from Dako/Agilent based on OTI1A4 and well-established system from Ventana/Roche based on D5F3 were most successful giving a pass rate of 100% and 95%, respectively. The mAb clone ALK1 was used by 8% of the participants with a pass rate 5%. A sufficient result in ALCL was seen but insufficient/false negative in the lung cancer emphasizing the need to re-optimize IHC protocols for ALK gene rearrangements in lung cancer
AMACR (p504s): 334 laboratories participated and obtained a pass rate of 93% (74% optimal) being on par to the level seen in all four NordiQC runs conducted for AMACR. Virtually identical pass rates were seen for LD assays typically based on the rmAb clone 13H4 and RTU systems based on either rmAb clone 13H4 or SP116. Efficient HIER preferable in an alkaline buffer and careful calibration of the primary Ab were the two main prerequisites for an optimal staining result.
BAP1: 163 laboratories participated in this first assessment of BAP1 and obtained a pass rate of 69% (42% optimal). The focus for BAP1 as marker to differentiate reactive mesothelial lesions from malignant mesothelioma. At present no RTU systems available and all participants applied as such a laboratory developed (LD) assay. The mAb clone C-4 was most widely used and provided an optimal performance all main fully automated IHC platforms. 3-step detections systems were found to be main prerequisite for an optimal result.
CD30: 365 laboratories participated and obtained a pass rate of 75% (44% optimal). The well-established mAb clones Ber-H2 and JCM182 were used by 98% of all participants and applied as RTU systems in 70%. The mAb clone Ber-H2 was found to be most successful when applied in combination with a modified citric based low pH buffer and a 3-step detection system. The mAb clone JCM182 was also successful, but found challenging to calibrate for an optimal result due to an extended reaction in macrophages hampering the selective demonstration of e.g. Hodgkin cells.
CK5: 311 laboratories participated and obtained a pass rate of 71% (44% optimal). The pass rate was improved compared to 58% and 44% observed in the two latest assessments of CK5. As seen previously, the widely used mAb clone XM26 was significantly more successful compared to mAb clone D5/16 B4 with pass rates of 93% and 49%, respectively (pooled data for both concentrated and RTU formats). The mAb clone D5/16 B4 typically provided a too low analytical sensitivity. The RTU system from Ventana/Roche based on rmAb clone SP27 provided an impressive pass rate of 100%, 100% optimal. The improved pass rate in part contributed by harmonization of best practice protocol settings concerning clones, titers if concentrate and use of 3-step detection systems.
p53: 370 laboratories participated and obtained a pass rate of 55% (23% optimal) being an improvement from 44% in run 63 but still significantly inferior the level of 79% in run 38, 2013. This assessment focused on IHC for p53 to demonstrate different TP53 mutations in endometrial carcinomas. The widely used mAbs clones BP53-11 and DO-7 could both be used to provide optimal results providing the protocols were based on efficient HIER in an alkaline buffer and of central importance in combination with a 3-step detection system. As a consequence of the “new purpose of p53 IHC” the most important parameter being a careful calibration of the primary Ab to consistently identify p53 in low-level expressing structures as dispersed normal lymphocytes and stromal cells being of central importance in the diagnosis of carcinomas with loss of p53 expression. Due to the relatively low pass rate, p53 will be repeated in 2023, run 67 and reassessment will not be available.
Companion module - run C11
PD-L1 (TPS/CPS): 225 laboratories participated and a pass rate of 81% was observed. This was the first NordiQC assessment of PD-L1 for TPS/CPS status with focus on TNBCs in addition to NSCLCs. Similar to observations seen in previous runs, the insufficient PD-L1 IHC results were most frequently characterized by a too low TPS/CPS level changing PD-L1 status in one or more of the carcinomas included. The companion diagnostic PD-L1 IHC assay, 22C3 GE006, Dako/Agilent performed in concordance to the product guidelines, was most successful providing a pass rate of 97%. LD assays based on mAb clone 22C3 and performed on the fully automated platforms BenchMark and Omnis provided highly reproducible results with “an all-time high” pass rate of 95%.
PD-L1 (IC score): 141 laboratories participated and obtained a pass rate of 59%, being on par to run C10 and slightly reduced compared to the results in run C9. The assessment for PD-L1 IC score focused on the evaluation of the analytical accuracy of the PD-L1 IHC assays for urothelial carcinomas and TNBCs. The insufficient results were characterized by either pure false negative results (seen for SP142 based assays) or an extensive staining reaction in tumour compromising the evaluation of PD-L1 reaction in immune cells in combination with an either false negative or false positive result (seen for non-SP142 based assays). In concordance with previous runs, the Ventana/Roche PD-L1 SP142 IHC assays 741-4860 and 790-4860 were most successful providing a pass rate of 79% and 81%, respectively.
July 11th 2022
Søren Nielsen
Scheme director
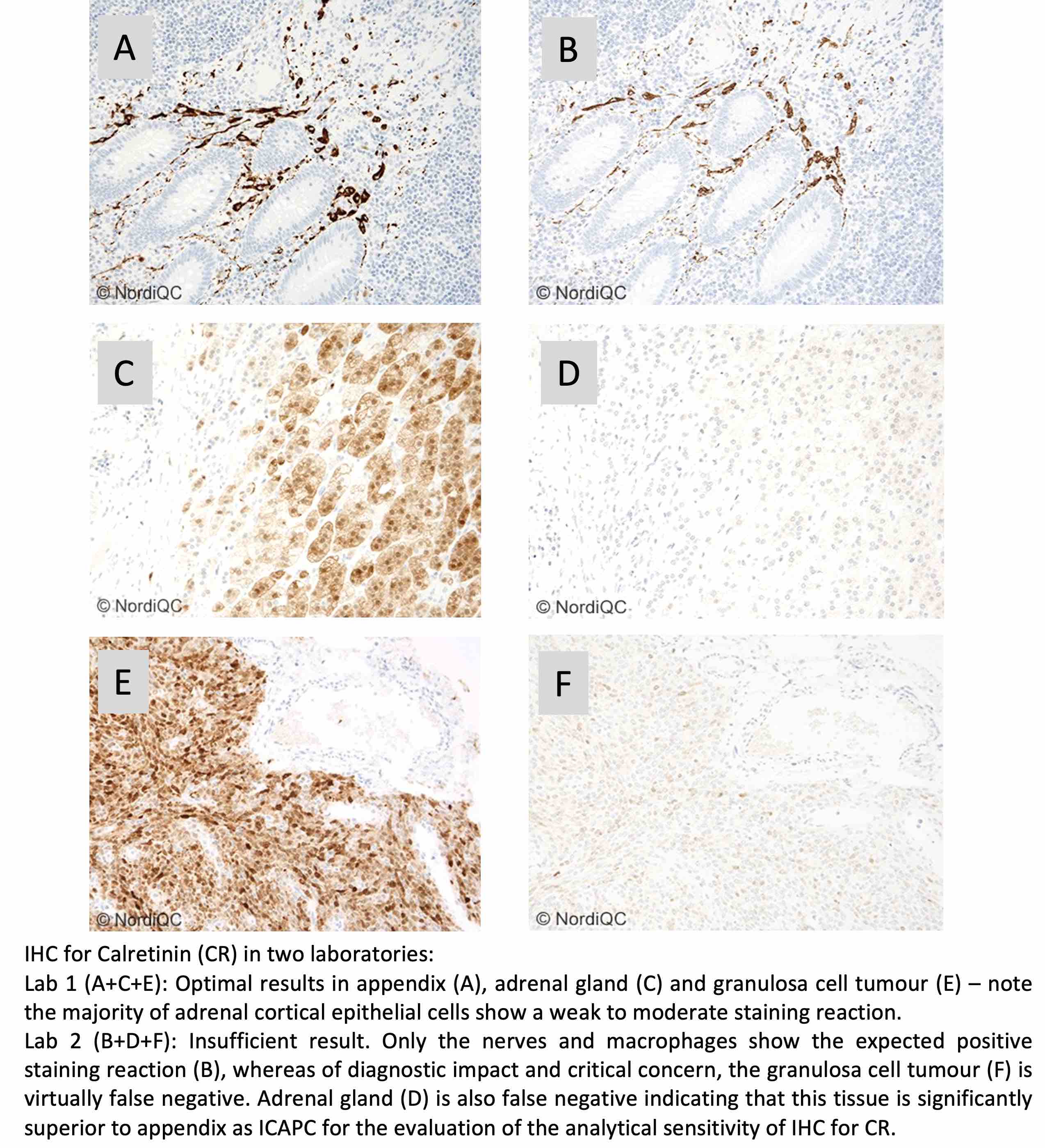
Run 64 (General module), B33 (Breast module) and H21 (HER2 ISH module) were accomplished from January to April 2021. In total, 517 laboratories participated in one or more of the three modules offered and more than 3.000 slides were assessed.
As usual, a short summary of the results is given below. Click on the epitope name to see the complete and detailed assessment report for the individual markers evaluated. In each assessment report, NordiQC intended to identify best practice methods and recommendable immunohistochemical critical assay performance controls (iCAPCs) to both guide the IHC protocol set-up and to monitor the IHC performance within the laboratories.
We encourage participants to update the submitted protocol settings and reagents used if changed after initial data entry. The data, including lot numbers, are used to analyze the overall performance and identify root causes for both inferior and optimal performance.
General module - run 64
CD56: 364 laboratories participated and obtained a pass rate of 72% (47% optimal). The pass rate increased slightly from the recent assessment run 61, 2021. Similar to all assessments for CD56, the take-home messages being usage of basic highly sensitive protocol settings and of utmost importance selection of a clone either as concentrate or RTU format capable to provide the expected results on the IHC platform used within in the laboratory.
CR: 339 laboratories participated and obtained a pass rate of 76% (50% optimal). In total eleven different Abs/clones could provide an optimal result. 80% of the participants used RTU formats. The Leica Biosystems RTU system based on clone CAL6 most successful followed by the Ventana/Roche system based on rmAb clone SP65. As concentrated format the mAb clone CAL6 provided superior performance and higher proportion of optimal results compared to the mAb clone DAK-Calret1, as the latter being inferior on fully automated systems as Dako Omnis and Ventana BenchMark.
DES: 370 laboratories participated and obtained a pass rate of 69% (41% optimal). The well-established and widely used mAb clones D33 and DE-R-11 were both very successful, providing being applied by appropriate protocol conditions. For mAb clone DE-R-11 application of HIER was found to be the main prerequisite for an optimal result being in contrast to the package insert for the Ventana/Roche RTU system recommending proteolysis providing a disappointing pass rate of 22%, none optimal. The Dako/Agilent RTU format IS/IR606 for Autostainer based on clone D33 was frequently migrated to Dako Omnis with an inferior result and also with an unacceptable pass rate of 22%.
PAX8: 337 laboratories participated and obtained a pass rate of 52% (27% optimal). The pass rate has now improved slightly from the low level of 37% in run 56, 2019. In concordance to the previous assessments the rmAbs clones SP348, ZR-1 and mAb clone BC12 gave encouraging results. In contrast, the widely used mAb clone MRQ-50 provided a poor performance especially on the Ventana BenchMark and Dako Omnis platforms giving a too low analytical sensitivity and at the same time it also gave a cross-reaction to other PAX subtypes as PAX5 in B-cells. Due to the relatively low pass rate, PAX8 will be repeated in 2023 and reassessment will not be available.
SATB2: 173 laboratories participated in this second assessment of SATB2 and obtained a pass rate of 75% (42% optimal) being an improvement from 58% in run 58, 2020. The majority of participants used the rmAb clone EP281 and was found to be more successful compared to other Abs as e.g., mAb clone SATBA4B10 and pAbs. Used within a LD assay, optimal results of EP281 could be obtained on all main fully automated IHC platforms especially when applied with HIER in an alkaline buffer and a 3-step detection system. LD assays and RTU formats/systems of EP281 provided similar performance.
Breast module - run B33
Estrogen receptor (ER): 407 laboratories participated and obtained a pass rate of 89% (54% optimal) being identical to the level seen in B32. rmAb clones SP1 and EP1 and mAb clone 6F11 could all be used to provide an optimal result. 86% of the participants used RTU systems for the demonstration of ER and provided an overall pass rate of 91%. The RTU systems from Ventana/Roche based on clone SP1 and Dako/Agilent clone EP1 (Omnis), were most successful with pass rates of 96% and 97%, respectively, when used by vendor recommended protocol settings. Too weak or false negative staining reactions were the predominant features of insufficient results. Tonsil was found to be recommendable as positive and negative tissue control.
Progesterone receptor (PR): 404 laboratories participated and obtained a pass rate of 91% (61% optimal) being on par to the level seen in the last three runs. mAb clones 16, PgR 636, PgR 1294 and rmAb clone 1E2 could all be used to provide an optimal result. 78% of the participants used RTU systems from Ventana/Roche, Dako/Agilent and Leica Biosystems and in total obtained a pass rate of 93% when applying these assays as “plug-and-play”. The Leica Biosystems’ RTU system based on clone 16 was most successful with 100% optimal results. In this assessment, a false negative staining reaction was the predominant feature of the insufficient results, but false positive results were also observed. Tonsil and uterine cervix in combination were found to be recommendable negative and positive tissue controls.
HER2 IHC: 384 laboratories participated and obtained a pass rate of 82% (54% optimal) almost identical to the latest run B32 and slightly reduced compared to the level seen in the three previous assessment runs B29-B31. In this assessment, the CE-IVD approved HER2 IHC assay PATHWAY®, Ventana/Roche and the recently launched HercepTest™ Dako/Agilent for Omnis were most successful providing a pass rate of 93% and 100%, respectively, however PATHWAY® with a higher proportion of optimal results of 79% compared to 35% for HercepTest™, Dako Omnis. LD assays based on a concentrate or RTU with no predictive claim were used by 22% of participants giving a pass rate of 71%. The insufficient results were primarily characterized by false negative results or a poor signal-to-noise ratio compromising the read-out.
HER2 ISH module - run H21
HER2 ISH: 154 laboratories participated in the technical assessment of HER2 ISH (BRISH) and obtained a pass rate of 56% (45% optimal) and the level declined in most recent assessments. The VENTANA HER2 Dual ISH DNA Probe Cocktail, 800-6043 and INFORM™ HER2 Dual ISH assay, 780-4422/800-4422 were used by 93% of all participants. As observed in virtually all latest assessments, the insufficient results were mainly caused by large negative areas and missing signals in >25% of the neoplastic cells in one or more of the samples included.
204 laboratories participated in the HER2 ISH scoring module (65 via FISH and 139 via BRISH) and, the consensus rate was 85%, and 86% for laboratories using FISH and BRISH, respectively.
April 22th 2022
Søren Nielsen,
Scheme director
